For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. By following this simple guide and utilizing the periodic table, you can efficiently find and convert atomic weights into molar masses for your chemical calculations and experiments. Design Copyright © 2017 Michael Dayah (). The same methodology can be applied to determine the molar mass of any element on the periodic table.Ĭalculating the molar mass of an element is crucial in chemistry, as it provides a bridge between atomic-scale particles and macroscopic laboratory quantities. 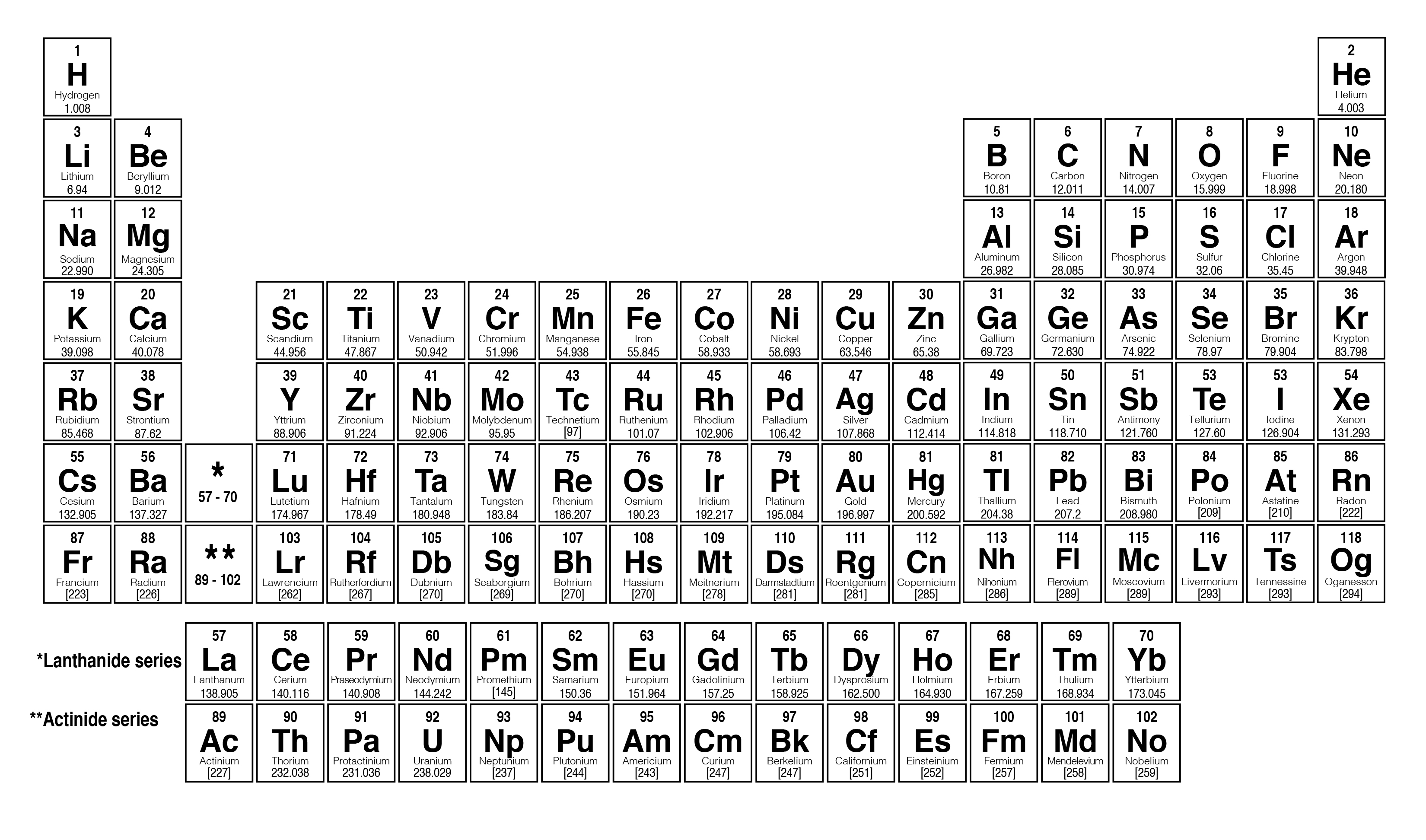
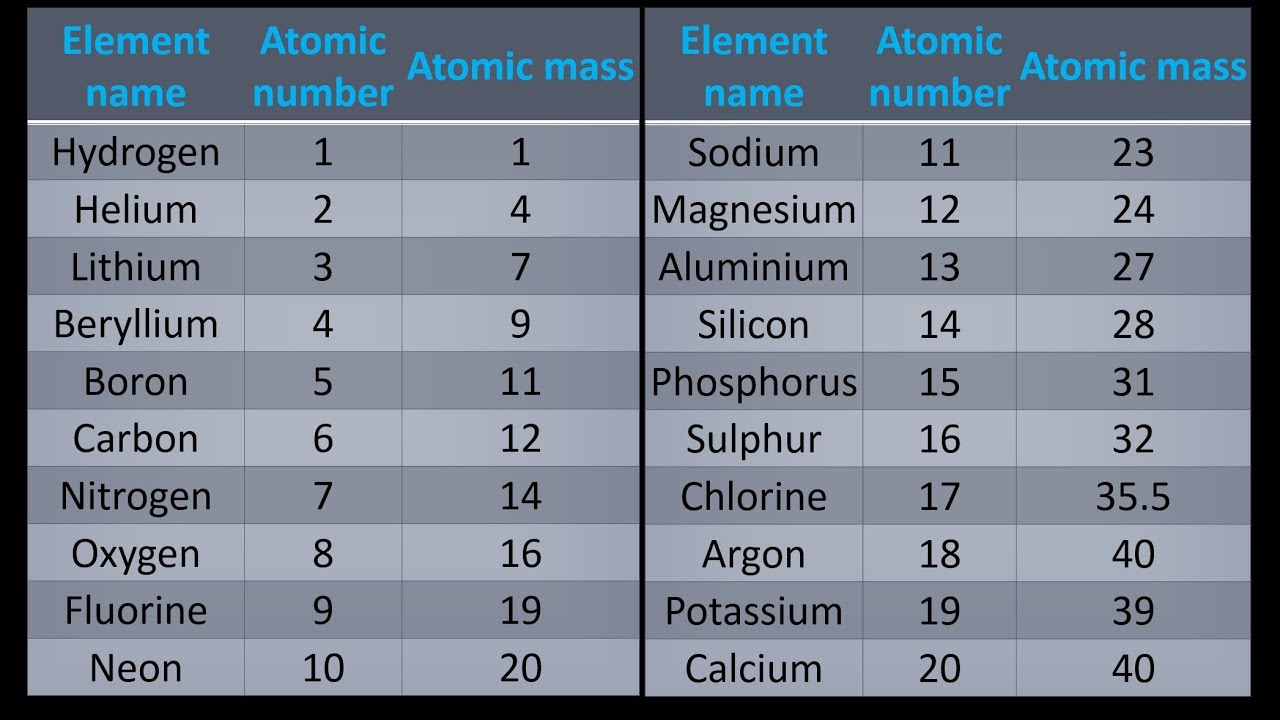
It is a moderately reactive metal and strong reducing agent. In our example of carbon, the molar mass would be 12.01 g/mol. Zinc has an electron configuration of Ar3d 10 4s 2 and is a member of the group 12 of the periodic table. To convert an element’s atomic weight into its molar mass, simply express it in g/mol. Convert atomic weight to molar mass: The unit for molar masses is grams per mole (g/mol). For carbon (C), for example, the atomic weight is 12.01.Ĥ. The atomic mass is a weighted average of all of the isotopes of that element, in which the mass of each isotope is multiplied by the abundance of that. On most periodic tables, this is a decimal number displayed below the chemical symbol. Find its atomic weight: Each element has its unique atomic weight (also known as atomic mass). Find your chosen element on the periodic table.ģ. Locate the element on the periodic table: The periodic table is arranged systematically and provides essential information about elements such as atomic numbers (number of protons) and atomic weights. Interactive periodic table with up-to-date element property data collected from authoritative sources. For example, if we want to find the molar mass of carbon, we can use the elemental symbol ‘C’.Ģ. Please note that the elements do not show. The unity for atomic mass is gram per mol. Identify the element: Determine the element you need to find molar mass for. The lightest chemical element is Hydrogen and the heaviest is Hassium. With over 4.89 million downloads, our popular PTE app is also available in web format. Make sure you enter the molecule of crystallization at last (e.g. Step-by-Step Guide to Calculating the Molar Mass of an Element:ġ. Indulge your curiosity with our complete periodic table of elements with masses, charges, chemical types, and everything else you can think of Our mobile app provides easy access to information any time, offline (for smartphones) and in full detail. Molar mass provides an easy way to convert between grams and moles for various elements and compounds. A mole is a unit for counting particles in chemistry, typically measured in terms of Avogadro’s number, which is 6.022 x 10^23 particles (atoms or molecules). Simply put, molar mass is the mass of one mole of a substance. In this article, we will delve deep into molar mass and discuss how to calculate it for any element. Molar mass plays a crucial role in chemistry, as it serves as a bridge connecting the microscopic world of atoms with the macroscopic world of chemicals in labs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
